PIROPLASMOSIS
Equine piroplasmosis is caused by the protozoan parasites Theileria equi (formerly Babesia equi) and Babesia caballi, which is approximately twice the size of T. equi. EP affects horses, donkeys, mules, and zebras and sometimes dogs. The wild zebra population is an important reservoir for the disease in Africa. Adult and nymphal ticks are capable of transmitting the disease.
While obtaining a blood meal from an infected horse, the tick ingests infected equine red blood cells. As the erythrocytes are digested in the tick’s digestive tract, parasite trophozoites are released and undergo sexual reproduction, resulting in the production of zygotes. Zygotes develop into sporozoites, which undergo division and spread via the tick’s haemolymph to its salivary glands. The causative agents are then transmitted when the infected tick bites a susceptible horse.
Genera of ticks that transmit EP agents include Dermacentor, Hyalomma, Rhipicephalus, and Boophilus. Approximately 15 species of ticks are capable of transmitting EP agents.
Once infected, a horse can take 7 to 22 days to show signs of illness. Clinical signs of acute EP are nonspecific, and mimic many other diseases and conditions. Signs observed are the result of red blood cell destruction, complement activation, and release of inflammatory mediators (including bradykinin, histamine, and 5-hydroxytryptamine). The severity of clinical signs reflects the number of cells destroyed and the degree of activation of the complement and inflammatory cascades.
Treatment via medication is instituted in affected horses. Diminazene diaceturate, phenamidine isethionate, and amicarbalide diisethionate are effective in eliminating clinical signs of B. caballi infection. Buparvaquone and other antitheilericidal drugs demonstrate some efficacy against T. equi, and may eliminate the parasite when combined with imidocarb. Theileria equi is more refractory to treatment than B. caballi, and higher dosages of imidocarb are required.
Because EP agents are transmitted by ticks, tick control is a vital preventive measure. Infected horses must be quarantined and isolated to reduce exposure to ticks. Pets and wildlife, including rodents must be prevented from entering the isolation area, as they may carry ticks capable of transmitting EP agents. No vaccine is available.
THEILERIASIS
Theileriasis, also called east coast fever, is a tick born disease of cattle, sheep and goats caused by protozoan blood parasites, Theileria annulata, Theileria parva and Theileria ovis of the Genus Theileria, Family Theileriidae and Order Piroplasmida.
Animals are infected through the bite of vector ticks.
Ticks possess large numbers of infective parasite Sporozoites, which develop in the salivary glands during the first two to four days of engorgement of nymph or adult ticks. The first visible stages are found in lymphocytes and the infected lymph nodes shows an increase in mitotic figures. Within next several days parasites are increasingly apparent in the local nodes and elsewhere in the lymphoid and reticulo-endothelial tissue.
Clinical signs include loss of appetite, dullness, depression, high temperature, salivation, lacrimation, difficulties in breathing, anaemia, haemoglobinuria, jaundice, convulsion and death. Other symptoms include, enlargement of lymph glands, pale mucous membrane and nodules in the skin, enlargement of liver, spleen, kidney and gall bladder and presence of necrotic ulcers in the abomasums of stomach.
Diagnosis involves microscopic examination of blood with Giemsa’s stain, presence of punched necrotic ulcers in abomasum of dead animal and serological test
Treatment is done by administering Oxytetracycline, Berenil, Antihistamins and Multivitamins.
TRYPANOSOMIASIS
Animal trypanosomiases include the following: Chagas; Nagana or Animal African trypanosomiasis, also called ‘Souma’ or ‘Soumaya’ in Sudan; Surra; Mal decaderas of central South America; Murrina de caderas of Panama; Dourine; Cachexial fevers; Gambian horse sickness of central Africa; Baleri of Sudan;
Kaodzera or Rhodesian trypanosomiasis; Tahaga of camels in Algeria; Galziekte or bilious fever of cattle or gall sickness of South Africa and Peste-boba of Venezuela.
African animal trypanosomiasis (AAT) is a disease complex caused by tsetse-fly-transmitted Trypanosoma congolense, T. vivax, or T. brucei in cattle but can cause serious losses in pigs, camels, goats, and sheep. Infection of cattle results in acute or chronic disease characterized by intermittent fever, anemia, occasional diarrhea, and rapid loss of condition and often terminates in death. In southern Africa the disease is widely known as nagana, which is derived from a Zulu term meaning “to be in low or depressed spirits”— a very apt description of the disease.
Ruminants are widely known to be active reservoirs of the trypanosomes. Wild Equidae, lions, 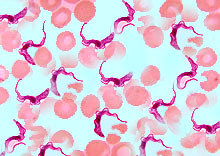 leopards, and wild pigs are all susceptible and can also serve as carriers of trypanosomes. Of the three African animal trypanosomes, only T. vivax occurs in the
leopards, and wild pigs are all susceptible and can also serve as carriers of trypanosomes. Of the three African animal trypanosomes, only T. vivax occurs in the
western Hemisphere in at least 10 countries in the Caribbean and South and Central America.
In Africa, the primary vector for T. congolense, T. vivax, and T. brucei is the tsetse fly. These trypanosomes replicate in the tsetse fly and are transmitted through saliva when the fly feeds on animals. The three main species of tsetse flies for transmission of trypanosomes are Glossina morsitans, which favors the open woodland of the savanna; G. palpalis, which prefers the shaded habitat immediately adjacent to rivers and lakes; and G. fusca, which favors the high, dense forest areas. Trypanosomiasis is also mechanically transmitted by other biting flies of the genera Tabanus, Haematopota, Liperosia, Stomoxys and Chrysops flies.
Initial replication of trypanosomes is at the site of inoculation in the skin, where swelling and a sore is caused. Trypanosomes then spread to the lymph nodes and blood and continue to replicate. T. congolense localizes in the endothelial cells of small blood vessels and capillaries. T. brucei and T. vivax localize in tissue. Antibody does not clear the infection, for the trypanosome has genes that can code for many different surface-coat glycoproteins and change its surface glycoprotein to evade the antibody.
The cardinal clinical sign observed in AAT is anemia. Within a week of infection, there is usually a pronounced decrease in hemoglobin, red blood cell, and white blood cell levels, and within 2 months these may drop to below 50 percent of their pre-infection values. Also invariably present are intermittent fever, oedema and loss of condition.
In the early phases of infection, especially with T. vivax and T. congolense, the parasite can readily be observed by microscopic examination of a wet-mount of blood slides. Thick blood films and stained with Giemsa are also a good technique.
Fly eradication and drug prophylaxis are effective trypanosomiasis control methods. Some of the older drugs such as the quinapyramine derivatives Antrycide and AntrycideProsalt are still used and give effective protection against T. brucei infection in horses, camels, and cattle. The drug pyrithidium bromide is useful in the prophylaxis of T. vivax and T. congolense infections in cattle, sheep, and goats and can give protection for up to 6 months.
The most widely used of the newer drugs is isometamidium chloride. Homidium bromide has also been found to be an effective drug in Kenya, and the newly introduced arsenical Cymelarsan is effective in treatment of T. brucei infection. A very widely used drug is diminazine aceturate, which is effective against all three African animal trypanosomes. The isometamidium drugs are also excellent chemotherapeutic agents as are the quaternary ammonium compounds Antrycide, Ethidium and Prothidium.
No vaccine is currently available for African animal trypanosomiasis.
COCCIDIOSIS
Coccidiosis is the name given to a group of closely related diseases caused by a protozoan parasite called Eimeria, which develops inside cells lining the intestine. As the parasites reproduce they cause bleeding and massive swelling of the gut. This leads to a huge loss of liquid and the bird is unable to absorb the nutrients from its food. Out of 11 species of Coccidia, the most common species are Eimeria tenella,E. acervulina and E. maxima, which cause chronic intestinal coccidiosis.
Eimeria tenella develops in the cells of the cecca which are the two blind sacs near the end of the intestine. It is one of the most pathogenic coccidia to infect chickens.
E. necatrix develops in the small intestine and later in the caecum, within deeper tissues of the small intestine and is a major pathogen of poultry.
E. accervulina and E. maxima develop in epithelial cells, primarily in the upper part of the small intestine.
The Sporozoite stage is the infective stage that contains special proteins and enzymes that allow it to penetrate the cell wall. Once inside the cell the sporozite begins its development through various stages. More parasites are produced during these stages, they in turn move on and in to healthy cells to begun the cycle again.
The result is weakened cell walls that allow moisture to leak and damaged blood vessels. This loss of blood and fluid is often fatal to the bird. Coccidiosis is passed to another bird during the oocyst stage, which is an egg-like structure that is excreted by the bird and can remain inactive for many weeks and still infect another bird. The infection starts when an oocyst is ingested. Once ingested, symptoms may appear as early as 4-6 days later.
Symptoms of the disease include listlessness, lethargy, weight loss and blood in the droppings or elsewhere in the body, diarrhoea, and dehydration are all common signs of Coccidiosis. The birds may also appear to have dirty or wet vents and may eventually die.
A controlled dose of coccidial vaccine is administered once to day-old chicks, initiating the development of natural immunity against Eimeria. Vaccination eliminates the need for withdrawal times, which are required when anticoccidials are used, as well as concerns about possible drug residues in poultry meat. Vaccination is also used to restore sensitivity to anticoccidials by replacing resistant, in-house strains of Eimeria with drug-sensitive Eimeria strains.
LEISHMANIASIS
Leishmaniasis is primarily a zoonotic disease in which wild and domestic animals such as the fox, jackal, rodents and wolves serve as reservoir hosts. Other animals in the surrounding areas can become infected and these are referred to as secondary or incidental hosts. The only proven vector of the Leishmania is the blood-sucking female sand fly of the genus Phlebotomus in the old world and Lutzomyia in the new world. Visceral leishmaniasis is caused by the parasites Leishmania donovani,Leishmania infantum and Leishmania archibaldi in the old world and by Leishmania chagasi in the new world..jpg)
The sand fly vector becomes infected when feeding on the blood of an infected individual or an animal reservoir host. The parasites live in the macrophages as round, non-motile amastigote fiorm (3-7 micrometers). The macrophages are ingested by the fly during the blood-meal and the amastigotes are released into the stomach of insect.
Almost immediately the amastigotes transform in to a motile, elongated (10-20 micrometers), flagellate promastigote form. The promastigotes then migrate to the alimentary tract of the fly, where they multiply by binary fission. Four to five days after feeding, the promastigotes move forward to the proboscis of the insect. When the sand fly next feeds on a mammalian host, its proboscis pierces the skin and saliva containing anti-coagulant is injected into the wound to prevent the blood from clotting, the promastigotes are transferred to the host along with the saliva.
Once in the host the promastigotes are taken up by the macrophages where they rapidly revert to the amastigote form. The leishmanias are able to resist the microbiocidal action of the acid hydrolases released from the lysozymes and so survive and multiply inside the macrophages, eventually leading to the death of the macrophages. The released amastigotes are taken up by the additional macrophages and so the cycle continues. Ultimately all the organs containing macrophages and phagocytes are infected, especially the spleen, liver and bone marrow.
Common symptoms include high undulating fever often with 2-3 peaks in 24 hours and drenching sweats which can easily be misdiagnosed as malaria; Chills, rigors, weight loss, fatigue, poor appetite, cough, burning feet, insomnia, abdominal pain, joint pain, anorexia, epistaxis and diarrhoea. Clinical sings include splenomegaly, hepatomegaly and lymphadenopathy.
The two pentavalent antimonial compounds, sodium stibogluconate and meglumine antimoniate have been used as first-line chemotherapeutic agents against all forms of leishmaniasis including visceral leishmaniasis. If treatment with pentavalent antimonials is unsuccessful a preparation of an aromatic diamidine, pentamidine isethionate (Pentamidine) or pentamidine dimethane sulphonate (Lomidine) is used. Amphotericin B, which is a macrolide, is another drug of second choice used in the treatment of leishmaniasis. There are three preparations of lipid associated Amphotericin B currently under trial for treatment of leishmaniasis: AmBisome (Vestar, USA), Amphocil (Liposome technology Inc, USA) and Amphotericin B lipid complex (Bristol Meyers Squibb, USA).